Congrats Adam on your Cell paper
Researchers create “Lipidomic Map,” offering insights into immunology
An international team of scientists has developed a method for simultaneously detecting thousands of lipid molecules that are displayed to T cells in the human immune system.
The study, co-led by Professor D. Branch Moody, MD, of the Division of Rheumatology, Immunity and Inflammation at Brigham and Women’s Hospital, and Adam Shahine, PhD, at the Monash Biomedicine Discovery Institute represents a collaboration among researchers from Oxford, United Kingdom, Monash University in Melbourne, Australia and Groningen, Netherlands. The results were published today in Cell.
The team developed a new and sensitive method to detect more than 2,000 lipids bound to CD1 antigen presenting molecules, which display antigens to the human immune system.
While scientists have long known that T cells recognise antigens, until the 1990s, it was thought that these antigens were always peptides derived from proteins. Because lipids are not encoded by genes and are instead made by enzymes and form into membranes, they have entirely different functions and positions in the cell.
The ability to measure many lipid antigens at one time will allow future researchers to cross-check any disease-related lipid of interest to the list of candidate lipid antigens from this map and potentially make connections to diseases.
Their efforts yielded the first integrated CD1 lipidomic map, which could help guide the investigation and discovery of lipid blockers and antigens for T cells and support the view that lipids normally influence immune responses.
The research builds on earlier methods that separate cellular lipids in one chromatographic system, which provided only a limited perspective. The new structural biology work, undertaken by Dr Shahine, ARC DECRA fellow, showed how lipids fit inside proteins using size-based mechanisms.
Combined, the structures and biochemistry detail rules about the size, shape, and chemical content of the kinds of lipids that can bind CD1 and cause a T cell response—either activation or deactivation. It is the latest in a series of studies that date back to the 1990s, when Brigham scientists discovered that T cells can recognise lipid antigens.
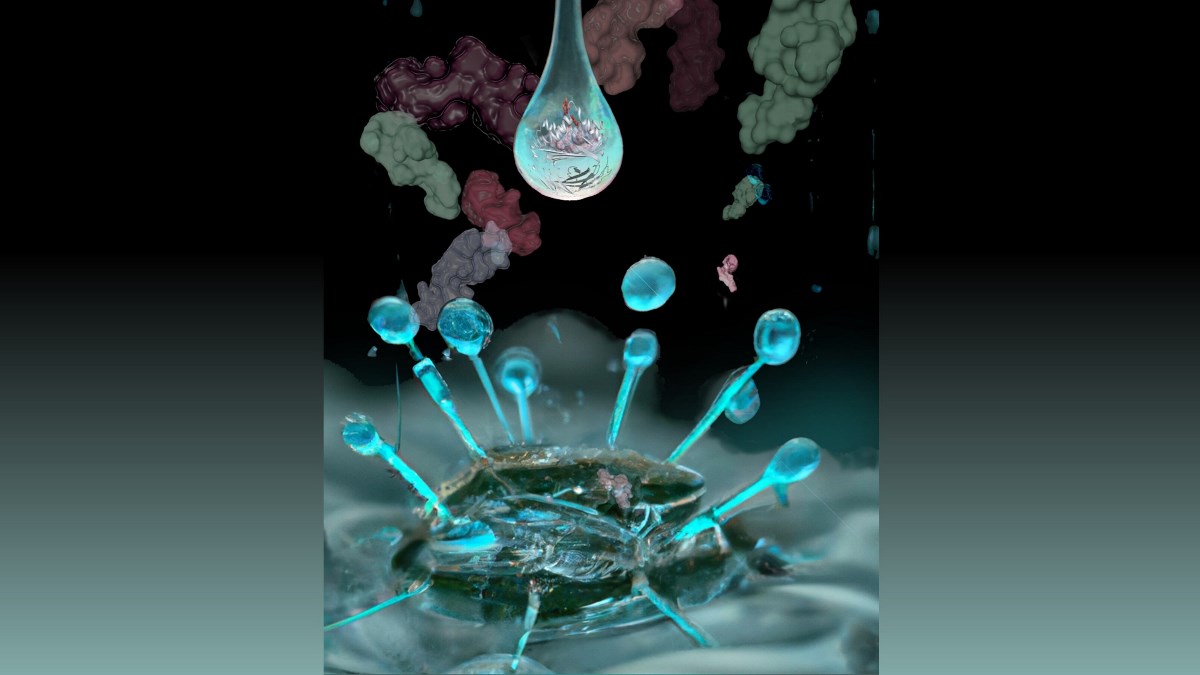
“In this ambitious decade-long, multidisciplinary study, we have characterized the full spectrum of cellular lipids that can be displayed to T cells. Further, we have collated 25 years of structural biology data, as well as new data collected at the ANSTO Australian Synchrotron, to standardize the rules that govern the molecular mechanisms in lipid presentation” said Dr Shahine. “Our hope is that the data generated in this study will serve as a foundation for future research in the field of lipid mediated immunity.”
Professor Moody said, “The Brigham provides an environment where physicians and scientists from differing fields can collaborate. This multidisciplinary effort involved biophysical techniques related to mass spectrometry and biological techniques related to lipid chemistry. The lipids informed immunological outputs, and the mode of lipid recognition is proven through X-ray crystallography.”
Read the full publication in Cell, titled CD1 lipidomes reveal lipid-binding motifs and size-based antigen-display mechanisms