Uncovering how T cells recognise the SARS-CoV-2 virus spike protein
The immune system is vitally important for resolving COVID-19 when individuals are infected with the SARS-CoV-2 virus. Moreover, the vaccines that are being administered to millions of people across the globe are designed to ‘pre-warn and arm’ the immune system so that if infected with SARS-CoV-2, individuals are significantly less likely to develop severe disease or die. Here, two crucial arms of the immune system, namely B cells and T cells, play a central role.
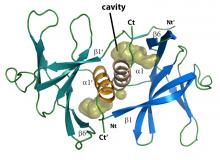
While we have a molecular understanding of how antibodies, which are produced by B cells, can bind and neutralise the spike protein from SARS-CoV-2, up until now researchers did not know how T cell receptors (TCRs), which are found on T cells, recognise antigens that arise from the spike protein.
“T cells play an important role in immunity against both SARS-CoV-2 vaccination and severe acute respiratory infection. Although T cells in COVID-19 have been studied previously, the molecular basis underpinning TCR recognition of SARS-CoV-2 remained unknown. It has been a pleasure working with the Monash University team to conduct this extremely important work to understand how T cells recognise an antigen from SARS-CoV-2,” said University of Melbourne Professor Katherine Kedzierska, a laboratory head at the Peter Doherty Institute for Infection and Immunity.
In a world first finding, co-led by Monash University’s Dr Priyanka Chaurasia, Dr Jan Petersen and Professor Jamie Rossjohn, and Professor Kedzierska, the team analysed the TCR recognition of a spike protein fragment when presented by an immune molecule, termed Human Leukocyte Antigen A2 (HLA-A2). This work, which utilised the Australian Synchrotron, was published in the Journal of Biological Chemistry.
“This is a piece of a larger puzzle. While SARS-CoV-2 continues to evolve, we have to build our understanding of how effective immune responses work,” said Dr Jan Petersen.
The team provided important molecular insight into understanding how T cells of the human immune system respond to SARS-CoV-2. Different individuals mount differing immune responses to SARS-CoV-2, and this work provided fundamental insight into such an immune response.
Read the publication in the Journal of Biological Chemistry titled Structural basis of biased T cell receptor recognition of an immunodominant HLA-A2 epitope of the SARS-CoV-2 spike protein
[Paper: J Biol Chem. 2021 Aug 10;297(3):101065. doi: 10.1016/j.jbc.2021.101065.]
Also featured in ANSTO article: Understanding how adaptive immune cells recognise and interact with the SARS CoV-2 virus